25 September 2019
 Our October edition of Research Round-up has collected three very different advances in our understanding of human organ systems.
Our October edition of Research Round-up has collected three very different advances in our understanding of human organ systems.
In the Nature article “Conserved cell types with divergent features in human versus mouse cortex,” Hodge et al. analyzed the cellular composition of the human cortex at the single nucleus RNA level.
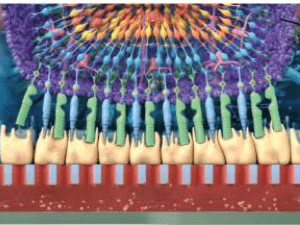
In the eLIFE paper “Merging organoid and organ-on-a-chip technology to generate complex multi-layer tissue models in a human Retina-on-a-Chip platform,” Achberger et al. demonstrate the creation of a sophisticated retina-on-a-chip with vasculature-like perfusion, which can recreate drug-induced retinopathy.
Gleiberman et al. describe the development of a microfluidic pancreatic islet-on-a-chip to accelerate efforts to study and develop effective treatments for diabetes, in their latest paper “Synchronized stimulation and continuous insulin sensing in a microfluidic human Islet on a Chip designed for scalable manufacturing.”
Mapping the human brain
We may have come one step closer to understanding the crucial differences in mammalian brain function thanks to research from the Allen Institute for Brain Science in Seattle, together with their collaborators, published in the Nature article “Conserved cell types with divergent features in human versus mouse cortex” on 5 September, 2019. This ambitious project set out to classify the cell types in the human cortex – the brain region associated with higher cognitive abilities and the most complex structure known to biology. The researchers made use of post-mortem tissue and tissue taken during neurosurgery for epilepsy resections – analyzing a total of 15,928 nuclei from the middle temporal gyrus (MTG) of the cortex of eight donor brains. The team used single nucleus RNA sequencing to analyze gene expression revealing 75 transcriptomically distinct cell types, representing non-neuronal, excitatory and inhibitory cells, which broadly mapped to data from previous studies but also revealed several additional neuron types.
The study went on to use these data to define the architecture of the MTG, finding that excitatory neuron subtypes segregated by layer, suggesting that anatomical location alone is not sufficient to predict neuronal cell type. In terms of non-neuronal cells, the study identified two astrocyte types with different distributions, which were distinguishable by their gene expression and morphology.
To compare cellular architecture between mouse and human, the authors aligned the human MTG transcriptomics with mouse primary visual cortex and mouse anterior lateral motor cortex. While transcriptomic organisation appeared conserved between species, divergences of homologous cell types was apparent at the single gene and gross structural level – differences that the authors claim will be “functionally relevant.” Importantly, this study revealed that serotonin receptors form one of the most divergent gene families between mouse and human, calling into question the utility and validity of mice for modeling neuropsychiatric disorders that involve serotonin signaling.
Overall, this exciting study indicates the basic transcriptomic similarity of cell types in mammalian brains but reveals vital differences in cell proportion and gene expression – differences that are likely to influence normal brain function, physiology and pathophysiology in humans compared to other mammals. This offers one more reason for the abject translational failures of preclinical studies using animals, and provides further evidence, if we needed it, that modeling the human brain will require more human approaches.
Modelling the human eye
The creation of a human retina system that may be used for testing new drugs and developing new treatments has been a challenge due to the great complexity of the human eye. The fact that animals do not share this same complexity or functionality, resulting in dismal translational failures, illustrates the urgent need for better models. Post-mortem retinal explants have been a useful resource, although the supply is limited. Additionally, these are not useful for studying retina development, and the variability between samples may be high due to inter-patient variability. Retina organoids produced with human induced pluripotent stem cells (hiPSC) are important tools, but they too miss essential features such as vascularization, some specific cell types, and the ability to replicate the physiological interplay of the many cell types in the human retina, specifically the photoreceptors and the retinal pigment epithelium (RPE). Thus, a new system capable of harbouring all the cells, and which replicates the complex physiology of the human retina, is urgently required.
In their eLife paper entitled “Merging organoid and organ-on-a-chip technology to generate complex multi-layer tissue models in a human Retina-on-a-Chip platform,” Prof. Dr. Peter Loskill’s group in Germany present a 3D in vitro system in which they combine different tissues created from hiPSCs to produce an ingenious retina-on-a-chip (RoC) model. The researchers used hiPSC to create a human retinal organoid and combined this with hiPSC-derived retinal pigment epithelium (RPE) to create a retina-on-a-chip with vasculature-like perfusion, which recapitulates the interaction of mature receptors with the RPE. In this new model, the photoreceptors can produce calcium flux and the RPE is able to do the phagocytosis of cell remains, which are vital functions of the human retina. When exposed to chloroquine and gentamicin, which are drugs known to cause retinopathies, the RoC developed the expected side-effects. This model can now be used to test many drugs and to hopefully help develop new treatments for diseases such as age-related macular degeneration, diabetic retinopathies and retinitis pigmentosa, which are the main causes of blindness in humans.
Measuring pancreatic islet function
Diabetes mellitus is a worldwide concern with increasing prevalence. Even though research on animals has been believed to play an important role in elucidating the pathogenesis of human diabetes and its complications such as retinopathy, nephropathy and neuropathy, all the animal models possess important limitations. Designing better, more human-relevant models that will allow precise identification of underlying disease mechanisms or to enable efficacy testing of therapeutic interventions is the need of the hour. Due to the failure of animal models in predicting the effectiveness of treatment strategies in clinical trials, suitable disease models are needed that can be used for studying pathogenesis, as well as for routine anti-diabetic drugs.
Pancreatic β cell function is usually assessed by measuring stimulated insulin secretion; this test is used to examine islet cell potency (from cadaveric or stem-cell derived sources) prior to transplantation. The measurement of glucose-stimulated insulin secretion requires manual liquid handling, heterogeneous stimulus delivery, and ELISA – all of which are extremely time- and resource-intensive, requiring many islet cells. These methods are still rampantly used, in spite of microfluidic devices that have been devised to address these limitations, mainly due to the steep learning curve for adopting these devices, and the fact that device materials are incompatible for large-scale manufacturing.
In their paper, “Synchronized stimulation and continuous insulin sensing in a microfluidic human Islet on a Chip designed for scalable manufacturing,” published in the journal Lab on a Chip, Glieberman et al. provide key insights into their work on designing and building a thermoplastic, microfluidic-based islet-on-a-chip that can be effectively scaled for manufacturing purposes and used feedback from potential end-users to make the chips more accessible, to enable wider adoption. Owing to its compatibility with commercial fabrication methods, islet loading, stimulation, and insulin sensing can all be automated with this novel system.
These chips are the first to offer automated, on-chip analysis of insulin release from human iselts. The authors developed a fluorescence, antibody-based on-chip assay which requires very small volumes (compared to ELISA) and which could be used to measure increases or decreases in insulin release and used this to show that individual islets could respond appropriately to glucose pulses. Further analysis indicated that perfusion and glucose stimulation of on-chip cultures did not adversely affect oxygenation or viability of the islets.
The authors concluded that the Islet-on-a-Chip could accelerate the efforts to study and develop effective treatments for diabetes by integrating scalable manufacturing materials, on-line continuous insulin measurement, and precise spatiotemporal stimulation, coupled with a design that is not complicated to use. Islet transplantation could be an effective treatment for diabetes but there are still significant issues regarding the prediction of success – including estimating islet function and ultimately calculating the size of transplant needed to restore function. This most recent advance provides a simple and sensitive method to measure dynamic islet function, and the authors “anticipate that it can help standardize and increase the predictive utility of islet testing.”
Image © Achberger et al., 2019 “Merging Organoid and Organ-on-a-Chip Technology to Generate Complex Multi-Layer Tissue Models in a Human Retina-on-a-Chip Platform.” eLife 8 (2019): e46188 made available under Attribution 4.0 International CC BY 4.0.

Post a comment